Learn about the launch of the Global Platform for Access to Childhood Cancer Medicines.
St. Jude Family of Websites
Explore our cutting edge research, world-class patient care, career opportunities and more.
St. Jude Children's Research Hospital Home

- Fundraising
St. Jude Family of Websites
Explore our cutting edge research, world-class patient care, career opportunities and more.
St. Jude Children's Research Hospital Home

- Fundraising
Global Platform for Access to Childhood Cancer Medicines



The Global Platform for Access to Childhood Cancer Medicines aims to provide an uninterrupted supply of quality-assured cancer medicines to low- and middle-income countries (LMICs).
It is estimated that 400,000 children develop cancer every year. Close to 90% of them live in LMICs, where survival rates are less than 30%. This is in stark contrast to high-income countries, where survival rates exceed 80%. A significant cause of this disparity in survival is insufficient access to quality-assured, safe, effective and affordable medicines for childhood cancer. A number of initiatives for improving access to cancer medicines have been developed over the past decade.
The Global Platform for Access to Childhood Cancer Medicines (Global Platform) will have the largest reach to children living with cancer of any program to date, anticipating expansion to provide medicines to treat approximately 120,000 children in LMICs.
St. Jude has committed to a six-year investment to launch the Global Platform in partnership with the World Health Organization (WHO), UNICEF and the Pan American Health Organization (PAHO) Strategic Fund.
The Challenge
An effective market for childhood cancer medicines is constrained by complex, interrelated systemic factors, resulting in market fragmentation, lack of availability of essential medicines, low-quality products, and high purchasing costs.
Availability
The availability of essential cancer medicines around the globe is inconsistent due to supply and demand issues and complicated regulations.

Quality
The quality of medicines is threatened when governments and administrations favor the lowest-cost bids.

Cost
The cost of childhood cancer medicines is often excluded from budgets, creating financial hardship for families in LMICs.
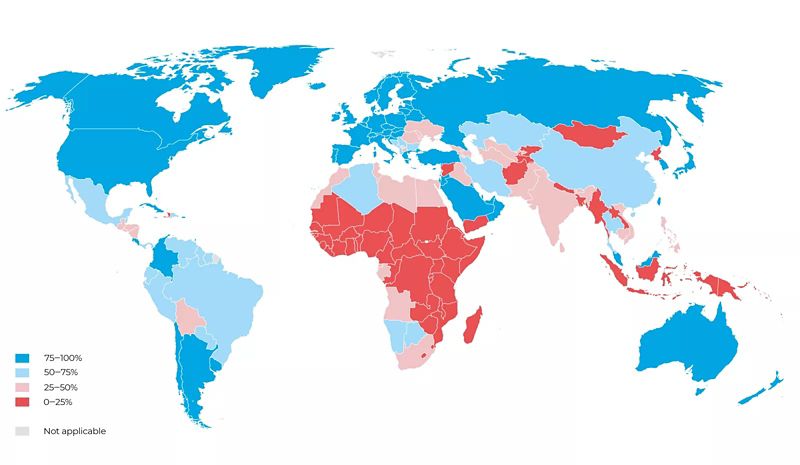
71% of low-income countries report general shortages in cancer medicines.

The Solution
The Global Platform brings together St. Jude, WHO, UNICEF, PAHO Strategic Fund, governments, the pharmaceutical industry, hospitals, non-governmental organizations, and nonprofit sector decision makers in a truly collaborative spirit to co-design a sustainable model. It will shape the market for childhood cancer medicines, rapidly increase access to care for thousands of children, and serve as a transformative model for the broader global health community in how we might work together to address these challenges in access to care for non-communicable diseases.
The Platform:
The Global Platform aims to provide an uninterrupted supply of quality-assured childhood cancer medicines with end-to-end support from selecting to distributing medicines according to the best possible care standards. The Global Platform will:
- Initially purchase and distribute medicines for childhood cancer at no cost to recipient countries.
- Distribute medicines of assured quality and efficacy. The list of medicines that the Global Platform will procure was selected through a rigorous process.
- Reinforce political commitment for childhood cancer control and support governments to safeguard families of children with cancer.
- Provide readiness support in advance of delivery of medicines, including assisting national stakeholders as they develop a tailored implementation roadmap.
- Take innovative approaches to shape the market for childhood cancer medicines to avoid interruptions in supply and promote timely regulatory approvals.
Read more about the WHO's involvement in the Global Platform.
The Impact:
Approximately 120,000 children in LMICs are anticipated to receive Global Platform medicines in the initial phases.
Frequently Asked Questions
Historical Resources

Panelists from St. Jude, WHO, SIOP & CCI speak about Global Platform for Access to Childhood Cancer Medicines.
View the Webinar
St. Jude and WHO address the challenge of access to childhood cancer medicines
Download an Overview
Download an information sheet on the Global Platform for Access to Childhood Cancer Medicines
Get the Facts
World Health Organization and St. Jude Children's Research Hospital to dramatically increase global access to childhood cancer medicines
Read the Press Release
WHO and St. Jude work together to expand access to childhood cancer medicines
Read the Article

