St. Jude Family of Websites
Explore our cutting edge research, world-class patient care, career opportunities and more.
St. Jude Children's Research Hospital Home

- Fundraising
St. Jude Family of Websites
Explore our cutting edge research, world-class patient care, career opportunities and more.
St. Jude Children's Research Hospital Home

- Fundraising
Registry
Other SJCARES Tools :

Overview
The SJCARES Registry tool is a hospital-based pediatric cancer registration and reporting system. Using an intuitive and secure cloud-based platform, the tool is designed specifically for low- and middle-income country contexts so that only relevant information is collected.
All data is owned by the individual member hospitals. St. Jude provides central quality control and assurance, along with easy reporting, to support population-based cancer registries.
SJCARES Registry provides opportunities to engage with and grow the registry and participate in global research.
Registry Tool
The Registry tool is a cloud-based solution for collecting pediatric hospital-based cancer registry data. The tool works with all major modern internet browsers and has proved functional in many countries, even with slow internet connections.
Watch a demonstration:
Why Use the Registry?
This is the first free, secure and thoughtfully crafted pediatric hospital-based cancer registry. It offers a standardized global platform, with only necessary data collected.
All data is owned by the individual member hospitals.
In addition:
- St. Jude can provide central quality control and quality assurance.
- There are opportunities to engage with and grow the registry together.
- The registry features easy reporting to support population-based cancer registries.
- There are chances to participate passively and actively in SJCARES global research.
Registry Content
The Registry tool set includes three registration modules that make up the global hospital-based childhood cancer network, with hospitals joining the SJCARES Registry within the six World Health Organization (WHO) regions, where the Core Registry is available now. The Extended Registry and Adverse Events Module will be launched soon.

The SJCARES Registry will grow as an institution's data needs and capacity for data entry grow.
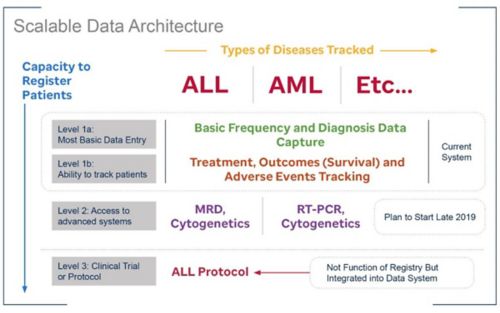
The tool was designed to support a hospital workflow in busy hospitals where data entry support is limited.
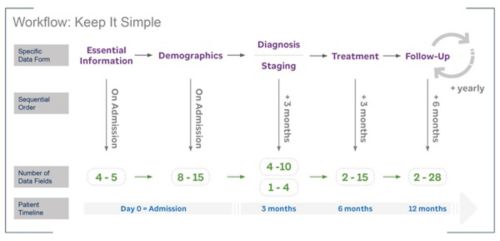
Responding to hospital registration needs, the first sibling registry, FEVEREG, is now available as part of the Registry ecosystem. This tool help with the surveillance of febrile episodes in low-and-middle-income country hospitals.
Data Ownership and Collection
All data is contractually owned by the individual member hospitals. Local registry teams can download their data at any time into one of several file formats for quality improvement or research purposes. St. Jude stipulates that any time the data is used for publication, all local ethics review procedures be followed.
SJCARES Registry provides opportunities for all St. Jude Global Alliance members to engage with registry data and participate in global research. As part of a data-use agreement, St. Jude will de-identify and store the data as part of the SJCARES data warehouse. Any investigator from a participating hospital may propose and conduct research that uses these de-identified data sets in accordance with the policies and procedures of the St. Jude Global Alliance. Whenever data from a hospital is used in a St. Jude Global Alliance study, a minimum of one investigator from the hospital will be eligible for co-authorship on any resulting publication.
Designed to Provide Global Standards
The SJCARES Registry:
- Is the product of a multidisciplinary effort with input from collaborators in every St. Jude Global Alliance region.
- Addresses a need for global standards to support pediatric hospital-based cancer registries.
- Allows hospitals to collect data to improve the quality of care at the local level and to share data with population-based registries at the national level.
The Registry team has taken an iterative design approach to ensure that the system meets the needs of users.
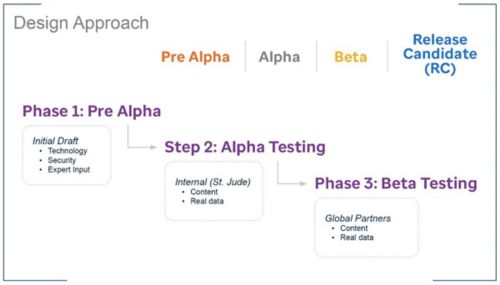
Careful consideration was given to ensuring that the data collected is the key data hospitals need for making, and tracking the impact of, important decisions. Examples include:
- ICD-O-3 coding and Toronto Consensus staging criteria
- Basic treatment data capture
- Follow-up and abandonment tracking
Data Security and Privacy
The physical servers for SJCARES Registry are securely maintained. Data is protected through a series of measures, including point-to-point encryption, data server encryption and data segmentation. St. Jude provides regular external audits of physical and logical security measures and practices in the data center. Protected health information is not viewable by St. Jude staff. The registry was built using OmniComm TrialMaster, an external third party.
SJCARES Registry uses industry best practices:
- Meets U.S. FDA and European GDPR requirements
- Uses the Microsoft Azure platform to manage identity and access
- Requires second-factor authentication by phone, email, text or application
St. Jude de-identifies the data in the SJCARES databank. Members can apply for data access to conduct a global or regional study. After the St. Jude Global Alliance steering committee reviews and approves the study, members are provided with access to the data and support for publication.
Registry Educational Curriculum
SJCARES Registry also provides educational resources.
Training Curriculum for Pediatric Cancer Registration
To provide a scalable solution for the initial and continuing education of learners who work with pediatric cancer registries in low- and middle-income country settings, the Registry team developed an asynchronous, competency-based education curriculum.
- A design-based research approach allowed for iterative cycles of revision and refinement of instructional material. This approach included:
- A scoping review of available cancer registration training materials
- Formulation of core competencies and learning objectives grouped hierarchically as modules and lesson plans to form a syllabus
- Development of course content and assessment questions through a three-stage iterative, peer-review process.
- A clinical research associate develops the initial content.
- Educators and pediatric oncologists review the content.
- A global panel of pediatric cancer registry experts provides feedback on the content.
- Breakdown of the cancer registration curriculum:
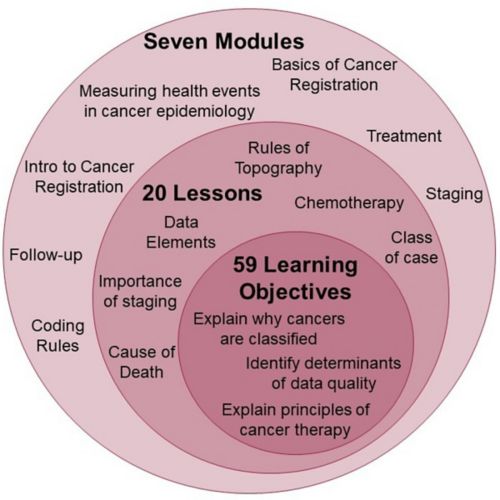
- Tutorial on effectively navigating the database system, TrialMaster
- Cancer registration course pathway
- Before accessing the registry, every user must complete the SJCARES Registry training in the onboarding process.
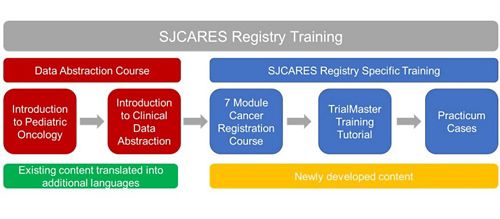
- A shorter version of the registry training is available for educational purposes only. This will be freely available.
- Completing this abbreviated version does NOT qualify a user to use the SJCARES Registry; to use the registry, users must go through the seven-step onboarding process.
Cure4Kids
Cure4Kids, an initiative of the St. Jude Children's Research Hospital Department of Global Pediatric Medicine, is an online resource for health care professionals around the globe who focus on life-threatening childhood diseases. Cure4Kids enables the web-based SJCARES Registry curriculum to be freely accessible to registered users.
Registry Training Specifics
MD Monitor (must be a physician)
- Data Abstraction Course, Intro to Pediatric Oncology Module: Optional
- Data Abstraction Course, Intro to Clinical Data Abstraction Module: Optional
- Cancer Registration Course: Required
- TM Training Tutorial: Required
Data Entry Specialist with medical (MD/RN) professional background
- Data Abstraction Course, Intro to Pediatric Oncology Module: Optional
- Data Abstraction Course, Intro to Clinical Data Abstraction Module: Required
- Cancer Registration Course: Required
- TM Training Tutorial: Required
Data Entry Specialist without medical background
- Data Abstraction Course, Intro to Pediatric Oncology Module: Required
- Data Abstraction Course, Intro to Clinical Data Abstraction Module: Required
- Cancer Registration Course: Required
- TM Training Tutorial: Required
Steps for Using the Registry
There are seven key steps for joining the registry platform.
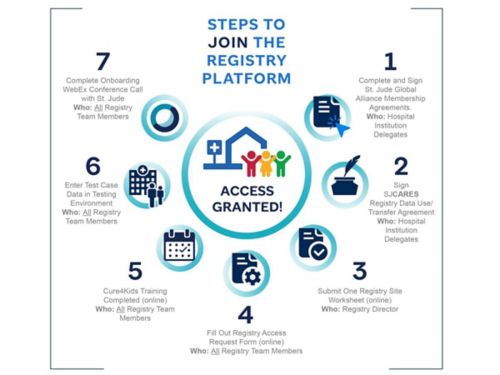
For Hospital Institution Delegates
1. Complete and sign the St. Jude Global Alliance membership agreement.
2. Sign the SJCARES Registry Data Use/Transfer Agreement.
For the Registry Director
3. Submit one registry site worksheet online.
For All Registry Team Members
4. Fill out the registry access request form online.
5. Complete the Cure4Kids training online.
6. Enter test case data in the testing environment.
7. Complete an onboarding WebEx conference call with St. Jude.
Registry Site Team Roles (English)
Función de roles del equipo del Registro
Our Team
Core Registry Development:
-
View Details
Nickhill Bhakta, MD, MPH
Associate Member, St. Jude Faculty
Chair, Regional Advisory Committee
Director, Disease Burden and Simulation
Co-Director, ARIA Program
Nickhill Bhakta, MD, MPH
Associate Member, St. Jude Faculty
Chair, Regional Advisory Committee
Director, Disease Burden and Simulation
Co-Director, ARIA Program
Affiliations
Research Interests
- Disease burden estimation, decision analysis, global epidemiology of childhood cancer
- Prioritization and the cost-effectiveness of childhood cancer treatment in low and middle income countries
- Pediatric leukemia, cancer survivorship
Contact Information
Nickhill Bhakta, MD, MPH
Global Pediatric Medicine
MS 721, Room S2013
St. Jude Children's Research Hospital
262 Danny Thomas Place
Memphis, TN 38105-3678
-
View Details
Carlos Rodriguez-Galindo, MD
Member, St. Jude Faculty
Executive Vice President
Chair, Department of Global Pediatric Medicine
Director, St. Jude Global
Co-Associate Director, Outreach, Comprehensive Cancer Center
Four Stars of Chicago Endowed Chair in International Pediatric Outreach
Carlos Rodriguez-Galindo, MD
Member, St. Jude Faculty
Executive Vice President
Chair, Department of Global Pediatric Medicine
Director, St. Jude Global
Co-Associate Director, Outreach, Comprehensive Cancer Center
Four Stars of Chicago Endowed Chair in International Pediatric Outreach
Affiliations
Research Interests
- Global medicine
- Pediatric solid tumors
Contact Information
Carlos Rodriguez-Galindo, MD
Global Pediatric Medicine
MS 721, Room S2015
St. Jude Children's Research Hospital
262 Danny Thomas Place
Memphis, TN 38105-3678
-
View Details
Paula Naidu, MPH, CCRP
Senior Director - Clinical Research
Affiliations
Short Biography
Prior to joining Global Pediatric Medicine, Paula helped build the clinical research infrastructure for the Department of Hematology as the clinical and experimental divisions joined to form a new department. She directed a team of study coordinators and research nurses to manage a clinical research portfolio which ranged from natural history of disease to multi-site, therapeutic trials. The scope of that work covered research concept to protocol completion: protocol development, regulatory compliance, data management, and quality assurance. She helped develop the clinical research career ladder at St. Jude, and continues to serve on the evaluation committee for the ladder. Paula has also served on the Internal Monitoring Committee, which is charged with oversight of the clinical research monitoring and auditing reports for the institution.
Paula graduated from the Honors Program at Auburn University, earning a Bachelor of Science in Biomedical Sciences. She later earned a Master of Public Health degree with a concentration in Epidemiology. She is a Certified Clinical Research Professional.
Contact Information
Paula Naidu, MPH, CCRP
St. Jude Children's Research Hospital
262 Danny Thomas Place, Memphis TN
-
View Details
Gia Ferrara, MSGH
Lead Clinical Research Associate
Affiliations
Short Biography
Gia Ferrara is a Lead Clinical Research Associate in the Department of Global Pediatric Medicine. She received her Bachelor of Science in Biology from Saint Mary’s College in Notre Dame, Indiana, where she conducted a senior comprehensive research project investigating the neurotoxicity of BPA in Sprague-Dawley rats. She then went on to receive her Master of Science in Global Health from the University of Notre Dame in July 2018. While at Notre Dame, she developed and conducted a study in collaboration with a partner organization in México investigating potential biomarkers for acute lymphoblastic leukemia.
Her experiences at Saint Mary’s and Notre Dame allowed her to combine her passions for traveling and serving others with her interests in science and medical research. Gia loves learning about and experiencing new cultures and is excited to serve as a clinical research associate in Global Pediatric Medicine at St. Jude.
Contact Information
Gia Ferrara, MSGH
St. Jude Children's Research Hospital
262 Danny Thomas Place
Memphis, TN 38105-3678
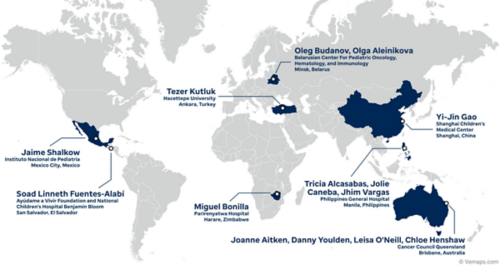
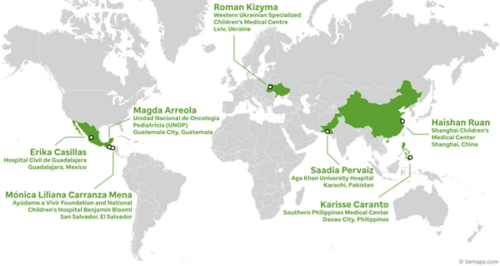
Beta Testing:
Phase I:
Phase II:
Registry Collaborative Working Group:
-
Nickhill Bhakta, MD, MPH
Director, Disease Burden and Simulation, St. Jude Global Pediatric Medicine
Memphis, Tennessee, USA
Nickhill Bhakta, MD, MPH
Associate Member, St. Jude Faculty
Director, Disease Burden and Simulation, St. Jude Global Pediatric Medicine
Memphis, Tennessee, USA
Affiliations
Research Interests
- Disease burden estimation, decision analysis, global epidemiology of childhood cancer
- Prioritization and the cost-effectiveness of childhood cancer treatment in low and middle income countries
- Pediatric leukemia, cancer survivorship
Contact Information
Nickhill Bhakta, MD, MPH
Global Pediatric Medicine
MS 721, Room S2013
St. Jude Children's Research Hospital
262 Danny Thomas Place
Memphis, TN 38105-3678
-
Joanne Aitken, BSc (Hon), MSc, PhD
Director, Australia Childhood Cancer Registry, Cancer Council Queensland
Brisbane, Australia
Joanne Aitken, BSc (Hon), MSc, PhD
Director, Australia Childhood Cancer Registry, Cancer Council Queensland
Brisbane, Australia
-
Lane Faughnan
Senior Coordinator, Research Operations, St. Jude Global Pediatric Medicine
Memphis, Tennessee, USA
Lane Faughnan
Senior Coordinator, Research Operations, St. Jude Global Pediatric Medicine
Memphis, Tennessee, USA
Affiliations
Contact Information
Lane Faughnan
Pathology
St. Jude Children's Research Hospital
262 Danny Thomas Place
Memphis, TN 38105-3678
-
Gia Ferrara, MSGH
Clinical Research Associate, SJCARES Registry, St. Jude Global Pediatric Medicine
Memphis, Tennessee, USA
Gia Ferrara, MSGH
Clinical Research Associate, SJCARES Registry, St. Jude Global Pediatric Medicine
Memphis, Tennessee, USA
Affiliations
Short Biography
Gia Ferrara is a Clinical Research Associate III in the Department of Global Pediatric Medicine. She received her Bachelor of Science in Biology from Saint Mary’s College in Notre Dame, Indiana, where she conducted a senior comprehensive research project investigating the neurotoxicity of BPA in Sprague-Dawley rats. She then went on to receive her Master of Science in Global Health from the University of Notre Dame in July 2018. While at Notre Dame, she developed and conducted a study in collaboration with a partner organization in México investigating potential biomarkers for acute lymphoblastic leukemia.
Her experiences at Saint Mary’s and Notre Dame allowed her to combine her passions for traveling and serving others with her interests in science and medical research. Gia loves learning about and experiencing new cultures and is excited to serve as a clinical research associate in Global Pediatric Medicine at St. Jude.
Contact Information
Gia Ferrara, MSGH
St. Jude Children's Research Hospital
262 Danny Thomas Place
Memphis, TN 38105-3678
-
Soad Fuentes de Aparicio, MD, MPH
Director, Mexico Region, St. Jude Global
Benjamin Bloom Children’s Hospital
San Salvador, El Salvador
Soad Fuentes de Aparicio, MD, MPH
Director, Mexico Region, St. Jude Global
Benjamin Bloom Children’s Hospital
San Salvador, El Salvador
Affiliations
Short Biography
Dr. Soad Fuentes-Alabi is a pediatric oncologist and global health expert with over 15 years of experience in advancing childhood cancer care in low- and middle-income countries (LMICs). She has played a pivotal role in strengthening health systems, implementing national childhood cancer control plans, and developing data-driven policies to improve pediatric cancer outcomes across Latin America.
She served as Scientific Director of the Pediatric Hematology-Oncology Day Clinic and the Population-Based Pediatric Cancer Registry (ROPESAL) at Fundación Ayúdame a Vivir (FAV) in Hospital Nacional de Niños Benjamin Bloom, El Salvador (2010-2025). During this time, she led critical initiatives in pediatric oncology, including the establishment of El Salvador’s first population-based pediatric cancer registry, contributing to regional cancer surveillance efforts and international research collaborations such as CONCORD-4.
Dr. Fuentes-Alabi earned her Doctor of Medicine (MD) from the Evangelical University of El Salvador, graduating as valedictorian, followed by a Pediatrics Residency at the National University of El Salvador and Hospital Nacional de Niños Benjamin Bloom. She completed her Pediatric Hematology/Oncology Fellowship at Universidad Autónoma de México and Instituto Nacional de Pediatría in 2006, where she was appointed Resident Coordinator for the Fellowship Program during her fifth year of training.
In 2016, she earned a Master of Public Health (MPH) in Clinical Effectiveness with a major in Epidemiology at Harvard T.H. Chan School of Public Health, supported by the St. Baldrick’s International Scholarship (2013-2018). Under the mentorship of Dr. Carlos Rodriguez-Galindo, Chair of the Department of Global Pediatric Medicine and Director of St. Jude Global, she developed a cancer epidemiology initiative in Central America aimed at establishing a population-based pediatric cancer registry in El Salvador, facilitating similar initiatives across Central America, and creating a childhood cancer epidemiology consortium to enhance data collection and research capacity in the region.
As a dedicated advocate for regional collaboration, Dr. Fuentes-Alabi has been an active member of the Asociación de Hemato-Oncología Pediátrica de Centroamérica (AHOPCA). She has spearheaded multi-country treatment protocols, including serving as Principal Investigator for the Neuroblastoma AHOPCA-2012 Protocol, and has been instrumental in promoting evidence-based pediatric cancer initiatives tailored to resource-limited settings.
Her expertise extends beyond clinical care, as she has led and advised multiple national pediatric cancer control strategies. In 2019, she was a Technical Committee Member for El Salvador’s National Plan for Pediatric Cancer Control, playing a key role in shaping national policies to ensure sustainable improvements in childhood cancer care.
From 2021 to 2024, she served as International Professional Consultant and Coordinator of the WHO Global Initiative for Childhood Cancer at the Pan American Health Organization (PAHO). In this role, she collaborated with United Nations organizations, including the International Atomic Energy Agency (IAEA) imPACT Review, and worked closely with Ministries of Health across Latin America to implement national cancer control plans, improve early diagnosis and treatment accessibility, and strengthen health system governance to ensure long-term sustainability.
Additionally, during her tenure at PAHO, she collaborated with multiple organizations to implement the WHO Global Initiative for Childhood Cancer (GICC) and its sponsor, St. Jude Children’s Research Hospital, through the St. Jude Global Pediatric Medicine Department and its multiple transversal programs.
-
Tezer Kutluk, MD, PhD, FAAP
Hacettepe University, Department of Medicine and Cancer Institute
Ankara, Turkey
Tezer Kutluk, MD, PhD, FAAP
Hacettepe University, Department of Medicine and Cancer Institute
Ankara, Turkey
-
Paula Naidu, MPH, CCRP
Director, Clinical Trials Management, St. Jude Global Pediatric Medicine
Memphis, Tennessee, USA
Paula Naidu, MPH, CCRP
Director, Clinical Trials Management, St. Jude Global Pediatric Medicine
Memphis, Tennessee, USA
Affiliations
Short Biography
Prior to joining Global Pediatric Medicine, Paula helped build the clinical research infrastructure for the Department of Hematology as the clinical and experimental divisions joined to form a new department. She directed a team of study coordinators and research nurses to manage a clinical research portfolio which ranged from natural history of disease to multi-site, therapeutic trials. The scope of that work covered research concept to protocol completion: protocol development, regulatory compliance, data management, and quality assurance. She helped develop the clinical research career ladder at St. Jude, and continues to serve on the evaluation committee for the ladder. Paula has also served on the Internal Monitoring Committee, which is charged with oversight of the clinical research monitoring and auditing reports for the institution.
Paula graduated from the Honors Program at Auburn University, earning a Bachelor of Science in Biomedical Sciences. She later earned a Master of Public Health degree with a concentration in Epidemiology. She is a Certified Clinical Research Professional.
Contact Information
Paula Naidu, MPH, CCRP
St. Jude Children's Research Hospital
262 Danny Thomas Place, Memphis TN
-
Karina Braga Ribeiro, DDS, PhD
Santa Casa de São Paolo, Department of Medical Sciences and Public Health
São Paolo, Brazil
Karina Braga Ribeiro, DDS, PhD
Santa Casa de São Paolo, Department of Medical Sciences and Public Health
São Paolo, Brazil
-
Danny Youlden, BSc
Biostatistician, Australian Childhood Cancer Registry, Cancer Council Queensland
Brisbane, Australia
Danny Youlden, BSc
Biostatistician, Australian Childhood Cancer Registry, Cancer Council Queensland
Brisbane, Australia
-
Carlos Rodriguez-Galindo, MD
Chair, Global Pediatric Medicine, St. Jude Children’s Research Hospital
Memphis, Tennessee, USA
Carlos Rodriguez-Galindo, MD
Member, St. Jude Faculty
Chair, Global Pediatric Medicine, St. Jude Children’s Research Hospital
Memphis, Tennessee, USA
Affiliations
Research Interests
- Global medicine
- Pediatric solid tumors
Contact Information
Carlos Rodriguez-Galindo, MD
Global Pediatric Medicine
MS 721, Room S2015
St. Jude Children's Research Hospital
262 Danny Thomas Place
Memphis, TN 38105-3678












